About Us

Lifelong group is a diversified group with interests in manufacturing of auto components and medical devices and services in the areas of investments, education, and software & event management. The group’s annual revenues are in excess of $150 million. The Lifelong group is led by Mr. Atul Raheja GROUP -whose initial foray was a successful career in manufacturing and marketing of domestic appliances under the brand name of “Lifelong”. The company diversified into auto component manufacturing in 1995 by acquiring a small plastic injection molding company. From annual revenues of $0.5 million in 1995, today the business has grown to $150 million and caters to global auto majors such as Hero MotoCorp, General Motors, Arvin Meritor, Plastic Omnium etc.
Lifelong India, which is the auto component manufacturing company specializes in injection molding components and assemblies, aluminum pressure die casting, metal stampings and multiple commodity assemblies. Lifelong India is a key supplier to Hero MotoCorp (which is the world’s largest 2 wheeler company with revenues in excess of $3 billion) and GM and has also won GM’s global best supplier awards in various years. In addition, Lifelong India also supplies parts that get fitted in BMW, Audi, Ford, Skoda, Renault, Peugeot and Hyundai. Lifelong India manufactured components are exported to North America, South America, France, Spain, Italy and South Korea. Lifelong India has 2 manufacturing facilities, one in Daruhera – an industrial estate in the progressive state of Haryana and the other one in Haridwar, Uttarakhand.
In 2005, the Lifelong group further diversified into manufacturing and marketing of medical devices and disposables. Thus Lifelong Meditech Private Limited was created for manufacturing of Sterile Hypodermic Single Use Syringes, Sterile Hypodermic Single Use Needles and Infusion Sets. Lifelong Meditech Private Limited markets its products under Safeway & Lifelong brands and is the second largest producer of syringes in India with an installed capacity of 900 million syringes per annum.
Both the businesses – auto component manufacturing and medical devices are managed and run by professionals who have been hired from the best companies in the industry. The other companies under the Lifelong group are: Shadionline.com, which is a wedding management company; Refam Securities, which is an investment company; Lifelong Online Private Limited, which is the online platform for sales of household & personal care products; Lush Escapes, which is luxury travel management company. The Lifelong Group under the able leadership and vision of Mr. Atul Raheja has ambitious plans of growing the business. A large part of this growth will come from acquisitions and activities for which are already underway.

Lifelong Meditech Private Limited is engaged in manufacturing of Medical Disposable products. The products currently manufactured by Lifelong Meditech Private Limited are Sterile Hypodermic Single Use Syringes, Hypodermic Single Use Needles and Infusion Sets. Lifelong Meditech Private Limited markets its products under Safeway and Lifelong brands and it is the second largest producer of syringes in India with an installed capacity of 900 million syringes per annum. Though a large part of Lifelong Meditech’s sale comes from the international market but it is a prominent player in domestic market as well. The company has its strong presence in the Northern States of India such as UP, Punjab, Haryana, Rajasthan and Delhi and it is slowly increasing its presence in Western and South Indian states. In the international market, Lifelong Meditech exports its syringes across the globe with major presence in Latin America, Europe, CIS Countries, Middle East & Africa and South East Asia.
Lifelong Meditech which is a part of the Lifelong India group with more than 30 years of experience in Plastic Injection Molding deploys state of the art machinery to produce medical disposable products of utmost quality and precision. Our technical work force and their competence define our parameters of excellence. Lifelong Meditech employs close to 550 people at its Manesar Plant. The existing plant has been set up with the most advanced machines both in the moulding and clean room area. All the machines are fully automatic and most modern having state of the art technology. Production of syringes is being done under the able supervision of well qualified & experienced technicians & engineers who are carefully hired and trained.
We have a fully equipped laboratory and testing facility for continuous inspection and testing of the quality of raw materials & other inputs used in the manufacturing of syringes. Also our finished products undergo all tests as specified in the ISO standard for quality and safety. We strictly observe GMP guidelines in our manufacturing process and control quality at all stages of production. Each batch of our product is tested in our laboratory by experienced chemists using suitable chemical & biochemical methods. The production process quality control and working conditions in the factory are continuously supervised by qualified persons and get regularly audited by the state Drug Controller. We also have ISO, GMP and CE Certifications.
Since the last one year, Lifelong Meditech has successfully ventured into the realms of manufacturing, supplying and exporting of syringes to the developed world. The company has also set benchmarks of excellence by catering to the specific requirements of hospitals, healthcare institutions, clinical and science laboratories, pathology laboratories and public consumers. The company is on its path of consistent growth under a proficient professional leadership, since its inception in the year 2004. The profound knowledge and rich experience of the team has assisted the company to achieve a distinguished position in the market amidst the numerous syringes manufacturers in India.

Atul Raheja
Chairman Lifelong Group
Atul is a veteran with experience spanning over three decades in building and managing diverse enterprises specializing in domestic appliances, plastic injection molding, event management, training academy and investment management.
His plastic injection molding company caters to international automotive giants such as Hero MotoCorp, General Motors, Arvin Meritor, Plastic Omnium among others. He leads the Lifelong Group with an annual sales revenue of more than $150 million.
Atul is an industrial engineer by training and is at the helm of five more companies:
- Lifelong India, an auto component manufacturing company
- Shadionline.com, a wedding management company
- Refam Securities, an investment company
- Lifelong Online, an online business for appliances
- Lush Escapes, a luxury travel management company
An active of Rotary International, Atul is also a member of the CII Northern Regional Council.

Hamendra Srivastava
Director & Chief Executive Officer
Hamendra has three decades of professional experience working in the manufacturing industry. He started his career working with AF Ferguson, a leading audit and consulting firm and then worked with Samtel for 14 years where he handled various roles both in India and in Germany. For 3 years he worked as the CFO with Minda Huf, a flagship company of the Ashok Minda group which is a leading auto component manufacturer.
Since 2007, Hamendra has been with the Lifelong group and is the group CFO and responsible for business planning and budgeting, accounting, MIS and financial controls. In January 2010, he took over as the CEO of Lifelong Meditech In his role as the CEO. He is responsible for adding new products to our offerings, increasing capacities to meet demand, improving efficiencies in our manufacturing process to reduce costs and most importantly develop a professional organization capable of delivering the desired growth in business.
Hamendra is a Chartered Accountant by training and has been a thorough finance professional and has a vast experience in the areas of acquisitions, joint ventures and fund raising.

Jatin Raheja
Director Lifelong Online International LLC,
General Manager Business Development & Strategy
Lifelong Group
Jatin earned a dual degree in Industrial Engineering (B.S.) and Economics (B.A.) from University of California, Berkeley, United States of America. During his time there, he focused on supply chain management, discrete optimization, decision analysis, econometrics and international trade.
Prior to joining the Lifelong Group in 2018, Jatin worked at Cohesity, a tech start up in Silicon Valley, where he analysed data availability and reduced data loss between replication paradigms by using Monte Carlo Simulations. He then moved to Avalon, an Asia-focused strategy and management consulting firm. As a consultant he specialized in financial modelling, market entry strategy, business plan development and diversification strategy in the automotive, metals and renewable energy sectors.
Jatin Raheja is a Director at Lifelong Online International LLC and the General Manager of Business Development and Strategy at Lifelong Group. He has led and contributed to the following initiatives across the group companies:
- Lifelong India: Led an initiative to diversify the company to improve resilience and protect against market fluctuation by reducing dependence on a single market segment. Part of the team that developed and launched a new automotive component from a newly setup factory.
- Lifelong Meditech: Worked on the expansion strategy to reduce costs, improve efficiency and expand product portfolio for what will become one of the world’s largest syringe manufacturing units.
- Lifelong Online: Founded Lifelong Online International LLC, a subsidiary of the company in the UAE, to expand product reach to global markets starting with the US. Introduced cutting-edge IoT innovation to existing products.

Neeraj Kumar Singh
Sr. General Manager – Operations
Neeraj has three decades years of experience in managing large and complex manufacturing operations. He has worked with various medical device manufacturing companies both in India and overseas. Neeraj joined Lifelong Meditech in June 2010 and is an ace at project implementation, operations and maintenance. He is currently heading the entire manufacturing operations of Lifelong Meditech which includes projects, production, maintenance, sourcing, materials management and logistics.
Neeraj is experienced in the good manufacturing practices required in the manufacturing of medical devices. His skills in production planning and execution and efficient utilization of man, material and machinery enables the organization to produce goods at the optimum cost. Neeraj is an Electrical Engineer by training. He demonstrates excellent leadership skills by mentoring and motivating his 500 member operations team to drive production and derive maximum output at the highest efficiency levels.

Bhupinder Singh
General Manager – International Business
Bhupinder has over three decades of experience in the areas of International, domestic sales and marketing. His experience of fifteen years is with BPL SANYO limited, India a leading company manufacturing range of consumer durables, medical electronics and communications. He has an experience of working with International markets for over eight years. He has worked with Eastern Medikit limited which is a leading Healthcare Company in India and has large presence in International markets. In his last assignment with Medikit, he was heading Marketing team of Disposable Medical devices for South America and Europe.
Bhupinder joined Lifelong Meditech in May 2011 and heads Export Division. He is responsible for acquiring new customers and growing business in the international market. His other important responsibilities include product pricing and developing markets for new products. His expertise lies in strategy planning and execution in the areas of new business development, marketing and operations.
Bhupinder is a graduate in Science from University of Delhi, India and a diploma in Electronics. He was a active member of the Delhi flying Club and has attended various training programme in sales and marketing.

Inderjeet Singh
Asst. General Manager- Quality
lnderjeet Singh has 18 years of experience in Quality Management systems and expert of implementation techniques. He has an important responsibility of ensuring compliance to the quality measures outlined. He has established quality standards and implemented stringent quality control systems to continually improvement in quality of products as per international standards. Has worked with leading device manufacturing companies. lnderjeet Singh joined Lifelong in April, 2005 and has been responsible for defining and monitoring Quality policies, procedures and protocols essential to comply with the drug regulatory requirements.
lnderjeet Singh is well versed with documentation procedures and has successfully managed many international regulatory compliance audits such as ANVISA, INMETRO, GMP, ISO and CE and is aware of the applicable standards and guidelines. lnderjeet Singh is a B.Sc., LLB from CCS University, Meerut and Post Graduation Diploma in Drug Regulatory Affairs from Jamia Hamdard University New Delhi.
An organizations’ capabilities and intent are strongly reflected in the product it manufactures. In other words, the manufacturing competencies and facilities echo truly, the extent and the ability to manufacture products of the highest quality and safety standards for the best of the market it targets.
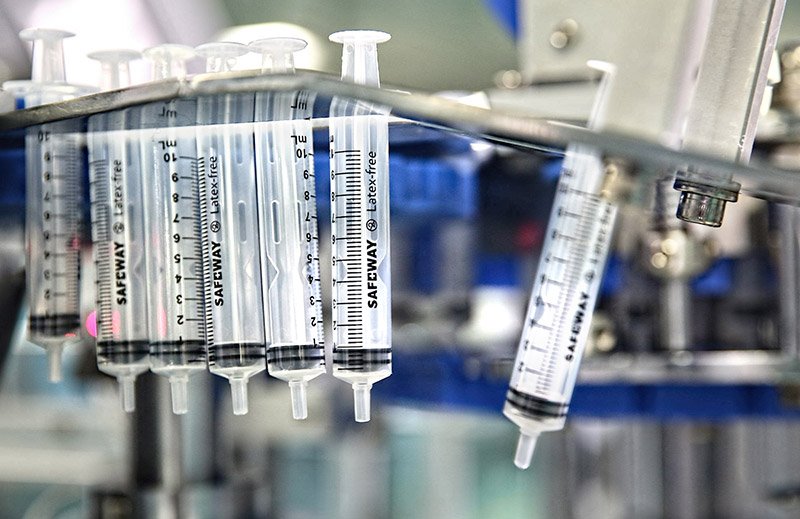
Lifelong Meditech Private Limited possesses the manufacturing strengths that have established it as a producer of world-class medical disposables and a major supplier of disposable syringes and needles to companies worldwide. Lifelong Meditech employs close to 550 people at its Manesar Plant. The existing plant has been set up with the most advanced machines both in the moulding and clean room area. All the machines are fully automatic and most modern having state of the art technology.
Below is a glimpse of our manufacturing facility:
Lifelong Meditech’s Quality System encompasses an exhaustive series of tests and inspection at different stages in the manufacturing cycle. Our manufacturing plant conforms to GMP and ISO standards. The discipline of this standard is evident in the physical ambience of our plant, the air and water quality, manufacturing technology, packaging and the sterilization process.
Special micro and HEPA filters at all air handling units ensure minimal contamination in the manufacturing areas. AH clean rooms are provided with three step change rooms and air showers at the entrance to ensure a dust-free environment. We have installed the latest technology automated machines in our plant. The superior assembly and packaging machines ensure repeatability and consistency in product quality.
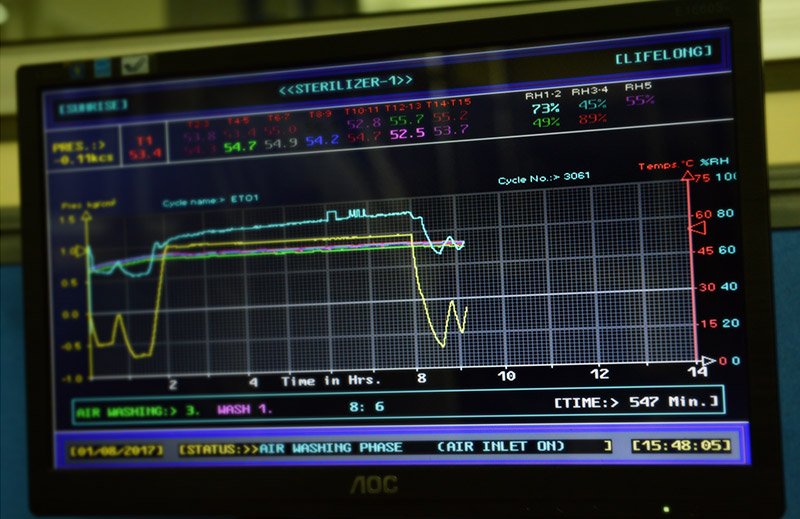
We use Ethylene Oxide gas to sterilize the products, as per 1SO 11135 standards. The entire process is automated and computerized. Each batch is well documented for the sterilization cycle and released for dispatch after written approval of a senior Quality Assurance official. Our products are packed to ensure minimal risk of contamination or damage during transportation.
Lifelong Meditech’s products comply with International Quality Standards, the testimony to this fact is the below certificates it has to its credit:




Lifelong Meditech has a well-defined Quality Policy and Quality Objectives which is as stated below.
Quality Policy
- Provide quality products at optimum cost
- Implement continuous improvement
- Maintain an effective quality management system which complies with regulatory requirement
- Ensure customer satisfaction and meet their requirements on sustained basis
Quality Objectives
- Achieve customer satisfaction through timely & cost effective supply of products of consistent quality
- Achieve continual improvement through:
- Employee training and motivation
- Productivity improvement
- Comply with all applicable Indian and International regulations

Quality Team
Global Reach

Contact Us
Registered Office:
Lifelong Meditech Private Limited
5th Floor, Unit No. 508, DLF South Court,
Saket District Centre, New Delhi - 110 017
Tel: +91-11-46017700 - 98
Fax: +91-11-46017799
Email: [email protected]
For International sales, please contact
Bhupinder Singh
Sr. General Manager - International Business
Mobile: +91 995 839 9899
Email: [email protected]
Factory:
Lifelong Meditech Private Limited
Plot No. 18, Sector - 5, IMT Manesar,
Gurgaon - 122 050, Haryana, India
Tel: +91-124-4406600
Fax: +91-124-4406699
Email: [email protected]
For operations, please contact
Neeraj Kumar Singh
Sr. General Manager - Operations
Mobile: +91 880 089 4406
Email: [email protected]
Corporate Film :
© 2022 Lifelong Meditech Private Limited.









































































































































.png)
.png)
.png)
.png)
.png)
.png)